1. Introduction
Saddle pulmonary embolism (SPE) refers to a thrombus located at the bifurcation of the main pulmonary artery, and is often associated with hemodynamic instability, increased complication rates, and poorer outcomes if not managed aggressively.1 Intracardiac thrombi (ICT), particularly within the left atrium (LA), left atrial appendage (LAA), or left ventricle (LV), significantly increase the risk of systemic thromboembolism, including ischemic stroke and acute ischemia involving abdominal organs or lower limbs.2 While echocardiography remains the most commonly used modality for diagnosing ICT, computed tomography pulmonary angiography (CTPA) is also valuable, particularly in certain clinical contexts.
We present the case of middle-aged obese male with SPE who developed transient neurological symptoms, later confirmed as an embolic stroke. Transthoracic echocardiography (TTE) was suboptimal due to body habitus, and a transoesophageal echocardiogram (TOE) was deemed high risk due to the need for uninterrupted anticoagulation in the context of a recent, high-burden PE. This limitation prompted a re-evaluation of the initial CTPA, which revealed an intracardiac thrombus in transit (ICTIT), seen as a filling defect in the right atrium (RA), extending across the interatrial septum (IAS) into the LA, findings consistent with thromboembolic material crossing through an atrial septal defect.
It is important to note that a Doppler ultrasound of the left leg was performed on admission and excluded the presence of DVT. However, a right leg Doppler could not be performed at that time due to the presence of an overlying cast.
2. Case Presentation
A middle-aged obese male with a history of reduced mobility following recent debulking surgery for a foot lipoma presented with hypoxic respiratory failure. CTPA revealed extensive submassive pulmonary emboli involving bilateral lobar, segmental, and subsegmental pulmonary arteries, along with significant right ventricular strain. The patient was commenced on weight-adjusted therapeutic subcutaneous low molecular weight heparin (LMWH) and admitted to the Respiratory Support Unit due to high oxygen requirements, which necessitated high-flow nasal cannula oxygen therapy.
Over the following 24 hours, his oxygen requirement steadily decreased, and he was successfully weaned to room air with concurrent improvement in dyspnoea and chest pain. He was subsequently transferred to the general respiratory ward. Shortly thereafter, he developed a sudden, transient episode of right homonymous inferior quadrantanopia and expressive dysphasia, both lasting approximately 15 minutes. Neurological examination was unremarkable following resolution of symptoms.
Neurology input was sought, and an MRI of the brain revealed multiple acute supratentorial infarcts consistent with a proximal embolic source. TTE was performed but proved inconclusive due to poor image quality, likely related to the patient’s body habitus. A TOE was deferred due to the risks associated with withholding anticoagulation in the context of a recent high-burden pulmonary embolism.
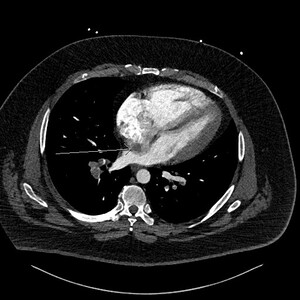
This prompted a re-evaluation of the initial CTPA by a specialist cardiothoracic radiologist. The following figures show selected images from the CTPA. Figure 1 is an axial slice demonstrating the already known bilateral filling defects within branches of the pulmonary arterial system, consistent with PEs (stars).
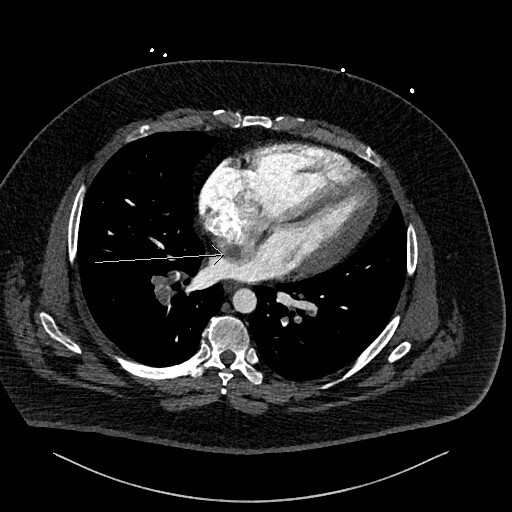
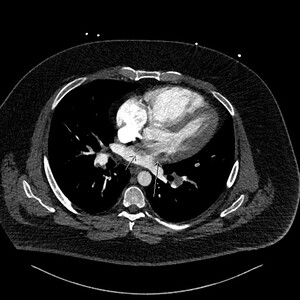
Figure 2 is an axial slice which shows a well-defined, round, homogenous and low attenuation filling defect (long white arrow) in the right atrium (RA) positioned adjacent to the interatrial septum (IAS). The filling defect is visible in the RA even despite the contrast mixing artefact in the RA chamber, the aforementioned features indicated the presence of a structure within the RA chamber, in this instance thrombus.
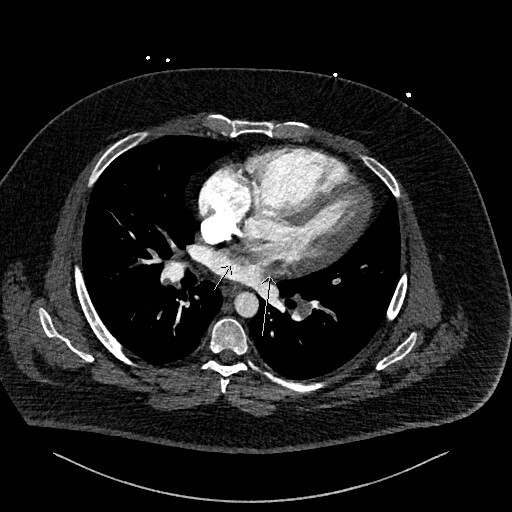
Figure 3 is the axial slice below and shows an elongated low attenuation and homogenous structure extending across a defect in the IAS into the left atrium (short arrow) and when viewed in contiguous slices was inseparable from the RA filling defect. This represented intracardiac thrombus in transit (ICTIT). There were further low attenuation, homogenous and well defined filling defects (medium white arrow) in the left atrium (LA). The radiologist also noted that the right heart chambers were enlarged compared to the left, and that both the IAS and interventricular septum (IVS) were inversely bowed toward the left – these are CTPA features that can indicate elevated right heart pressures and therefore right heart strain in the clinical context of PE.
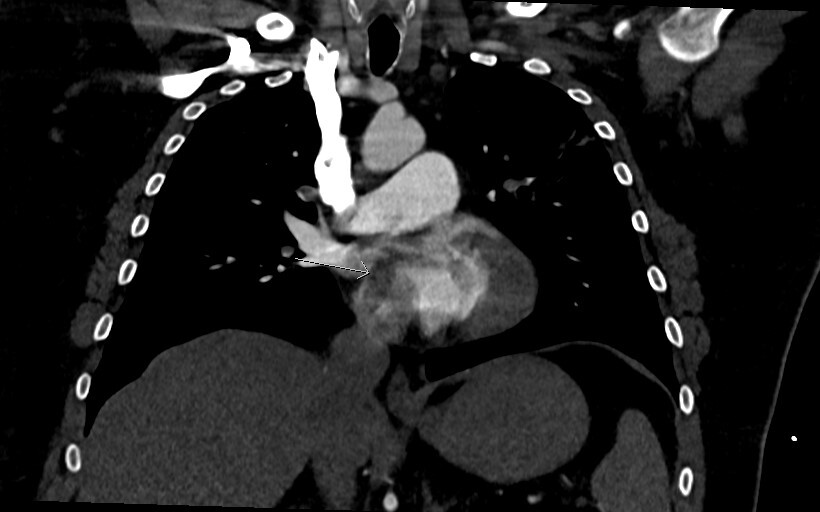
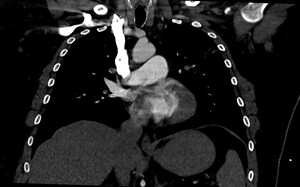
Figure 4 is a coronal image and shows the thrombus in transit again with a “boomerang” shape across the IAS (arrow heads).
The patient was maintained on therapeutic anticoagulation throughout his admission, initially with LMWH which was later switched to a direct oral anticoagulant (DOAC). He showed significant clinical improvement during this period. Multidisciplinary discussions involving the respiratory, radiology, and cardiology teams recommended continuation of therapeutic anticoagulation upon discharge.
A repeat CTPA performed at three months demonstrated complete resolution of the previously identified thrombus. A bubble TTE was subsequently conducted but proved inconclusive, failing to confirm the presence of an interatrial septal defect. At this stage, a TOE was considered feasible, and the patient was scheduled to undergo a TOE with a bubble study for further assessment.
Subsequent management, including consideration of defect closure, will be guided by the outcome of this investigation. During this period, the patient presented with new-onset right leg pain. Doppler ultrasound revealed a proximal right femoral vein DVT. It remained uncertain whether this represented a new thrombus or a persistent one, as the right leg Doppler had not been performed initially due to an overlying postoperative cast following debulking surgery. Anticoagulation was therefore transitioned to warfarin while awaiting the cardiology evaluation for TOE and potential interatrial septal defect closure.
3. Discussion
Our case illustrates a patient with SPE and ICTIT, straddling the IAS, identified on admission imaging, which subsequently resulted in an embolic stroke. PE with right heart thrombi are associated with significantly increased mortality, with 14-day mortality rates reported as almost double PE alone (21% vs 11%).3 Intracardiac thrombi located in the LA/LAA or LV substantially elevates the risk of systemic thromboembolism, including ischemic stroke and acute ischemia affecting abdominal organs and lower extremities.2
It is suggested that up to 18% of massive PE may also have concomitant right sided thrombi.4 Bi-atrial thrombi are particularly rare, and a meta-analysis of 174 patients with thrombus straddling a PFO suggested a 30-day mortality rate of 18.4%.5 Earlier recognition of ICTIT with biatrial thrombi on admission imaging may have altered the management strategy in this case. Fortunately, our patient did not sustain permanent neurological deficits, although this complication could have been anticipated had the thrombus been detected initially.
Currently, there is no consensus on the optimal treatment approach for PE with ICTIT. Therapeutic options include systemic thrombolysis, anticoagulation alone (using direct oral anticoagulants, warfarin, or low molecular weight heparin), or surgical/percutaneous embolectomy. Meta-analyses from 2002 and 2015 do suggest thrombolysis significantly improves outcomes compared to embolectomy and anticoagulation alone in patients with right heart thrombi.6,7 For thrombi which straddle a PFO, Myers et al report from meta-analysis that surgery was associated with the lowest 30 day mortality (10.8%), compared with thrombolysis (26.3%) and anticoagulation (25.6%).5
In this case, the long-term management decision was guided primarily by the presence of a recent embolic ischaemic stroke and the associated high risk of haemorrhagic transformation. While thrombolysis or catheter-directed therapies may be appropriate in selected cases of PE, these options were felt to carry a high bleeding risk in the context of recent cerebral infarction. Therefore, a conservative approach using therapeutic anticoagulation alone was deemed the safest option. This strategy aimed to balance the need to prevent further thromboembolic events while minimising the risk of intracranial haemorrhage.
Maffei et al discuss a case of PE with ICTIT. They decided for fibrinolysis in this case, with their patient also suffering a subsequent embolic stroke, requiring mechanical thrombectomy.8
Sattar et al describe a case of bi-atrial thrombus straddling a PFO in a patient who presented with features of an embolic stroke, lower limb DVT, large ICTIT and massive PE. Their patient reacted well to therapeutic dose LMWH and was subsequently warfarinised, showing complete resolution of ICTIT on follow-up TOE.9 Our patient likewise showed complete resolution of saddle PE and no evidence of ICTIT on follow-up CTPA, after receiving LMWH followed by DOAC.
However, our patient did suffer an embolic stroke after initiation of treatment. Similarly, Suenaga et al describe a case of PE, with right and left atrial thrombus present on CTPA images (rather than TTE images) suggestive of an ICTIT present across a PFO.10Their patient too was treated with therapeutic LMWH converted to DOAC, and also subsequently developed an embolic stroke (in this case, treated successfully with mechanical thrombectomy). Both cases highlight the importance of appreciation of bilateral atrial thrombi on acute CTPA imaging in PE and careful consideration of treatment approaches if ICTIT is identified. LMWH and DOAC present good treatment options for patients with PE alone, but we need further research to identify the best treatment approach, and the role of therapeutic LMWH and DOAC treatments, in patients with PE with ICTIT, to prevent subsequent embolic events.
Echocardiography remains the primary diagnostic tool for detecting intracardiac thrombi. Nuvvula et al describe a patient with PFO and ICTIT, presenting with stroke, saddle PE, and deep vein thrombosis (DVT).11 The authors suggest that TOE should be considered in all cases where there is concern for ICTIT, with inconclusive findings on TTE. However, TTE can be limited by suboptimal image quality due to factors such as patient body habitus or chronic pulmonary disease, as well as operator dependency. In our case, TOE was contraindicated due to the highrisk nature of withholding anticoagulation in the setting of acute pulmonary embolism. Our case in fact highlights the importance of carefully reviewing CTPA images for evidence of intracardiac thrombi, especially in patients with a large burden of pulmonary embolism when echocardiographic evaluation is limited or inconclusive.
4. Conclusion
This case highlights the diagnostic value of CTPA beyond assessment of the pulmonary vasculature, particularly in identifying intracardiac thrombus in transit. Careful review of CTPA images can reveal clinically significant findings that may not be detected on echocardiography, especially in acute high-risk pulmonary embolism where echocardiographic assessment may be limited or inconclusive. Recognition of these limitations is essential, as timely identification of intracardiac thrombus can influence management decisions and improve diagnostic clarity in complex presentations.
Conflict of Interest / Competing Interests
The authors declare no conflicts of interest.
Data Availability
No datasets were generated or analysed during the current study.
Ethics Approval and Consent
This case report was conducted in accordance with institutional and ethical standards. Written informed consent was obtained from the patient(s) for publication of this report and any accompanying images. Identifying information was removed or anonymized to protect patient privacy. Formal ethical approval was not required for this type of study.
Funding
This research received no external funding.
Reporting Guidelines
No formal reporting guideline was applicable to this study design
AI Tool Use Disclosure
Artificial intelligence tools were used solely for language editing and formatting. No artificial intelligence tools were used for data analysis, interpretation, or clinical decision-making. All content was reviewed, verified, and approved by the authors, in accordance with COPE guidelines on the responsible and ethical use of AI in scholarly publishing.
Preprint Disclosure
This manuscript has not been posted as a preprint.
Third-Party Material Permissions
All figures are original, and no third-party material requiring permission has been used.
AUTHOR CONTRIBUTIONS (CRediT)
Writing – original draft: Andrew Morrish (Equal), Fadwa Al-Qadi (Equal), Kanwal Tariq (Supporting). Writing – review & editing: Andrew Morrish (Equal), Fadwa Al-Qadi (Equal), Kanwal Tariq (Equal), Claudette Phillips (Supporting). Methodology: Andrew Morrish (Equal), Fadwa Al-Qadi (Equal). Resources: Andrew Morrish (Equal), Fadwa Al-Qadi (Equal). Supervision: Kanwal Tariq (Equal). Conceptualization: Kanwal Tariq (Supporting). Investigation: Claudette Phillips (Equal).

_image_showing_bil.jpeg)
_image_showing_bil.jpeg)